

Chemistry only
Alkenes are unsaturated hydrocarbons that contain a double carbon carbon covalent bond (C=C); this is the reactive functional group present in all alkene molecules. As soon as you see this functional group in any
molecule as a chemist you should think of only one thing- addition reactions! This carbon carbon double covalent bond (C=C) in an alkene molecule is vulnerable to attack by other substances which can simply add across it.
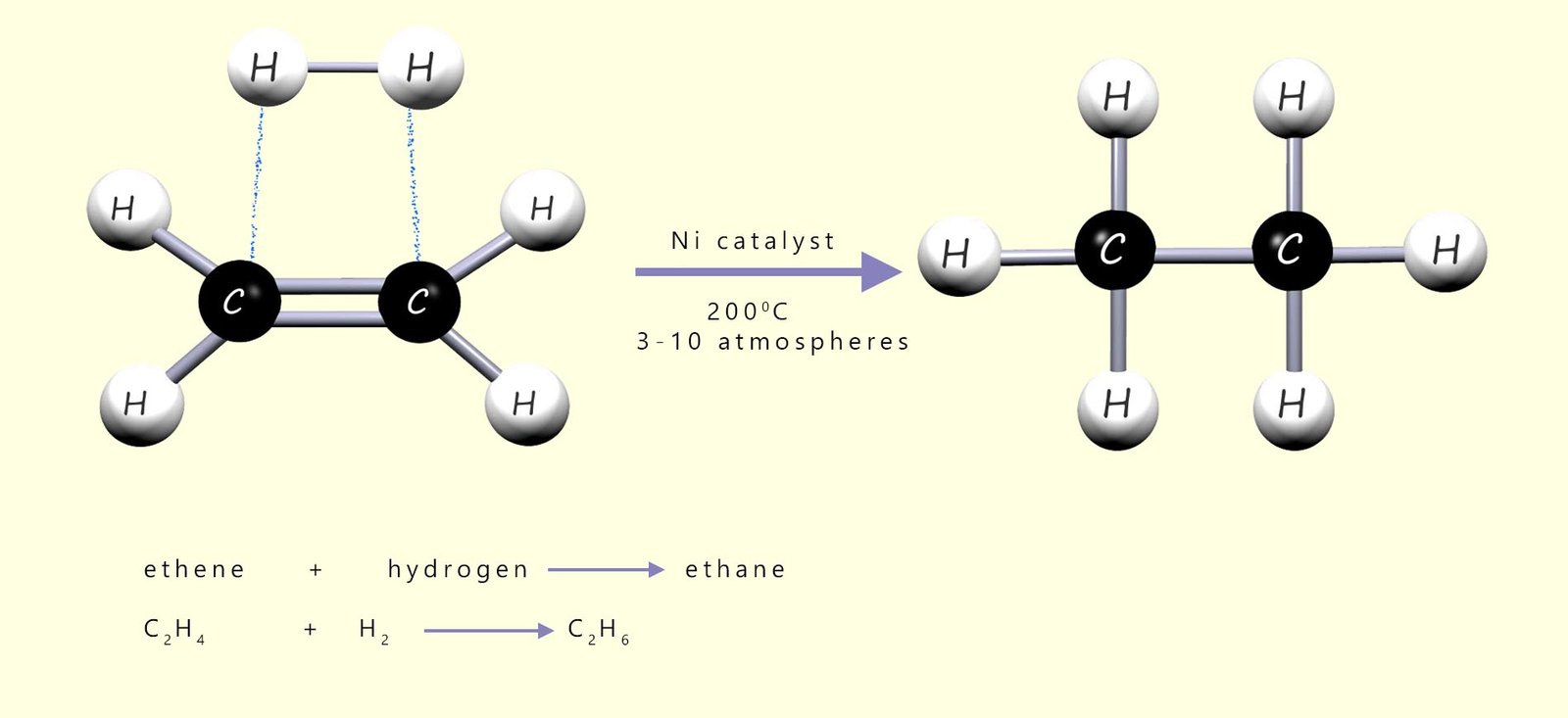
A nickel or platinum catalyst and a temperature of around 200 °C are required to carry out this hydrogenation reaction. The image below shows how a molecule of hydrogen gas (H2) can simply add across the reactive carbon-carbon double bond (C=C) in an unsaturated alkene molecule such as ethene to form a new saturated molecule of ethane gas.

One use of this hydrogenation reaction is in the manufacture of margarines. Margarine can be made from vegetable oils, for example rapeseed oil, sunflower oil or olive oil. However, spreading vegetable oil on your toast first thing in the morning is perhaps not a good idea unless you like soggy toast! Vegetable oils are often referred to as polyunsaturated molecules. This simply means that the long chain molecules found in a typical vegetable oil contain many carbon–carbon double bonds (C=C). During hydrogenation; hydrogen gas is added across some of these double bonds making the oil more saturated and turning it into a more solid product such as margarine.
The presence of lots of carbon-carbon double bonds (C=C) in a molecule lowers the melting point of the compound which is why many vegetable oils are liquids at room temperature. However by removing some or all of the sites of unsaturation in the vegetable oil its melting point can be raised. This means that the oil will now be a solid or perhaps a semi-solid if only some of the C=C bonds are saturated by the addition of hydrogen gas (H2) across them.

If all the carbon carbon double bonds (C=C) in the vegetable oil are removed by hydrogenation then the product will likely be a hard solid fat. However by hydrogenating only a specific number of the carbon carbon double bonds (C=C) present it is possible to determine just how hard and spreadable the margarine formed will be.
Ethene + hydrogen gas (H2) with a nickel catalyst, 200°C and 5 atmospheres pressure.
Which product is formed?
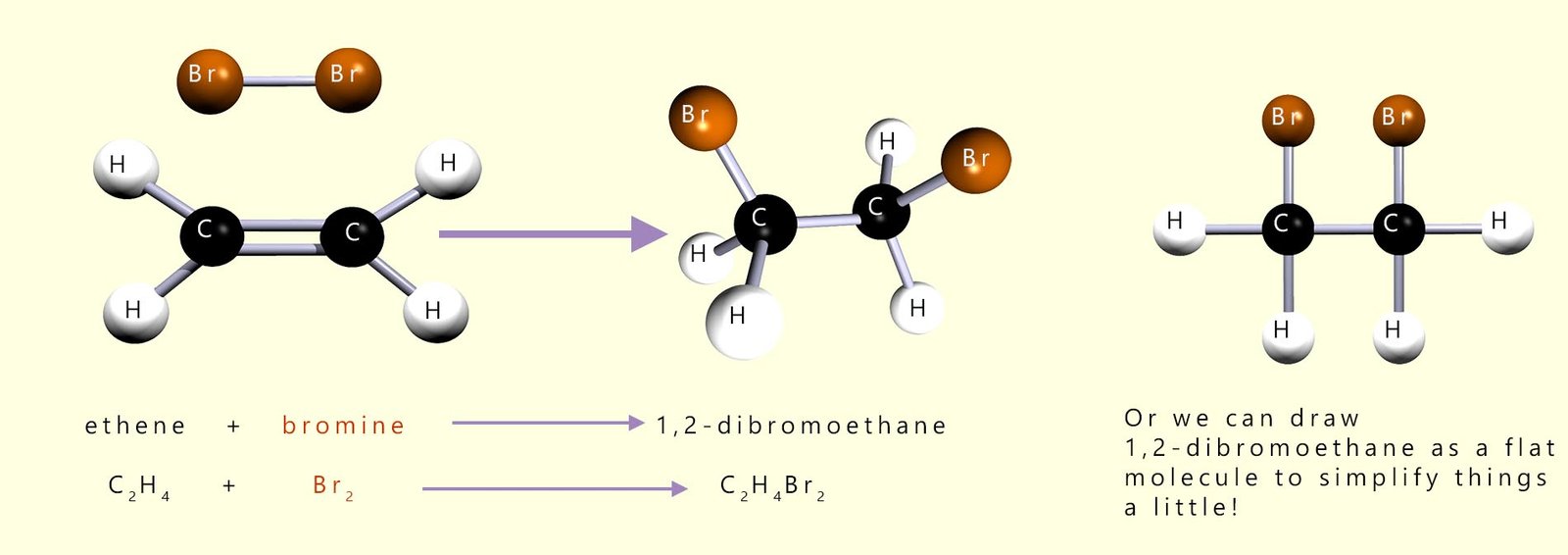
Other small molecules such as bromine and iodine will also add across the carbon carbon double covalent bond (C=C) to form saturated molecules. The diagram below show the products of the addition reaction of bromine to the unsaturated alkene ethene. Here a bromine molecule (Br2) can add across the double bond of an ethene molecule to form the colourless molecule 1,2-dibromoethane.
A similar addition reaction will occur if the bromine is swapped for chlorine (Cl2); here the chlorine molecule simply adds across the carbon-carbon double bond (C=C) in exactly the same way that the bromine molecule did, the product of the reaction this time will be 1,2-dichloroethane. Note the numbers 1,2- in the names of the compounds mentioned are simply used to indicate which carbon atoms the bromine or chlorine atoms have added to. Equations to show the addition of chlorine to ethene are outlined below: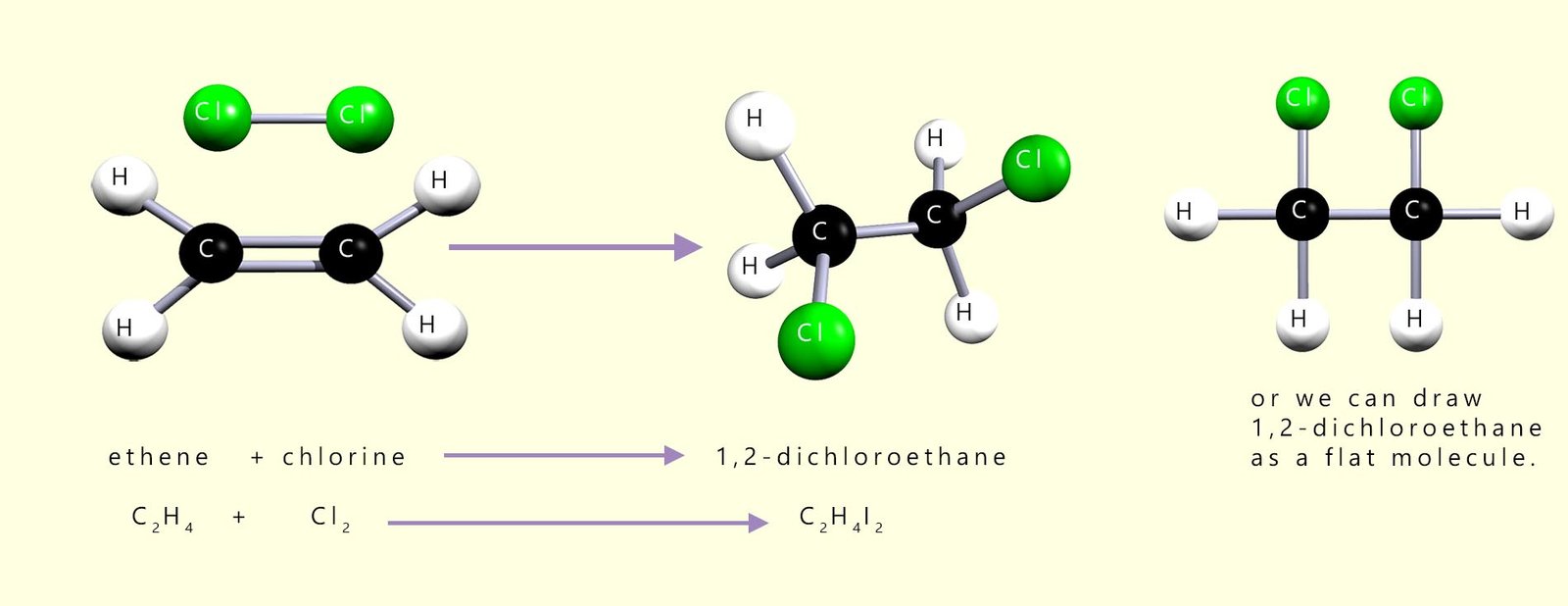
I am sure you would agree that the addition of a halogen (halogenation) such as chlorine or bromine across the carbon-carbon double bond (C=C) is very similar to the addition of hydrogen gas in the hydrogenation reaction above.
Ethene + bromine (Br2)
Which product is formed?
It is also possible to add steam across the C=C bond in alkenes to make alcohols. This method of making alcohols is called direct hydration. It requires a fairly high temperature of around 300 °C and a pressure of about 65 atmospheres as well as a phosphoric acid catalyst. The reaction is similar to the examples above in that a small molecule; in this case steam adds across the C=C bond in an alkene, this is shown below using the alkene ethene as an example, addition of steam to ethene will produce the alcohol ethanol:
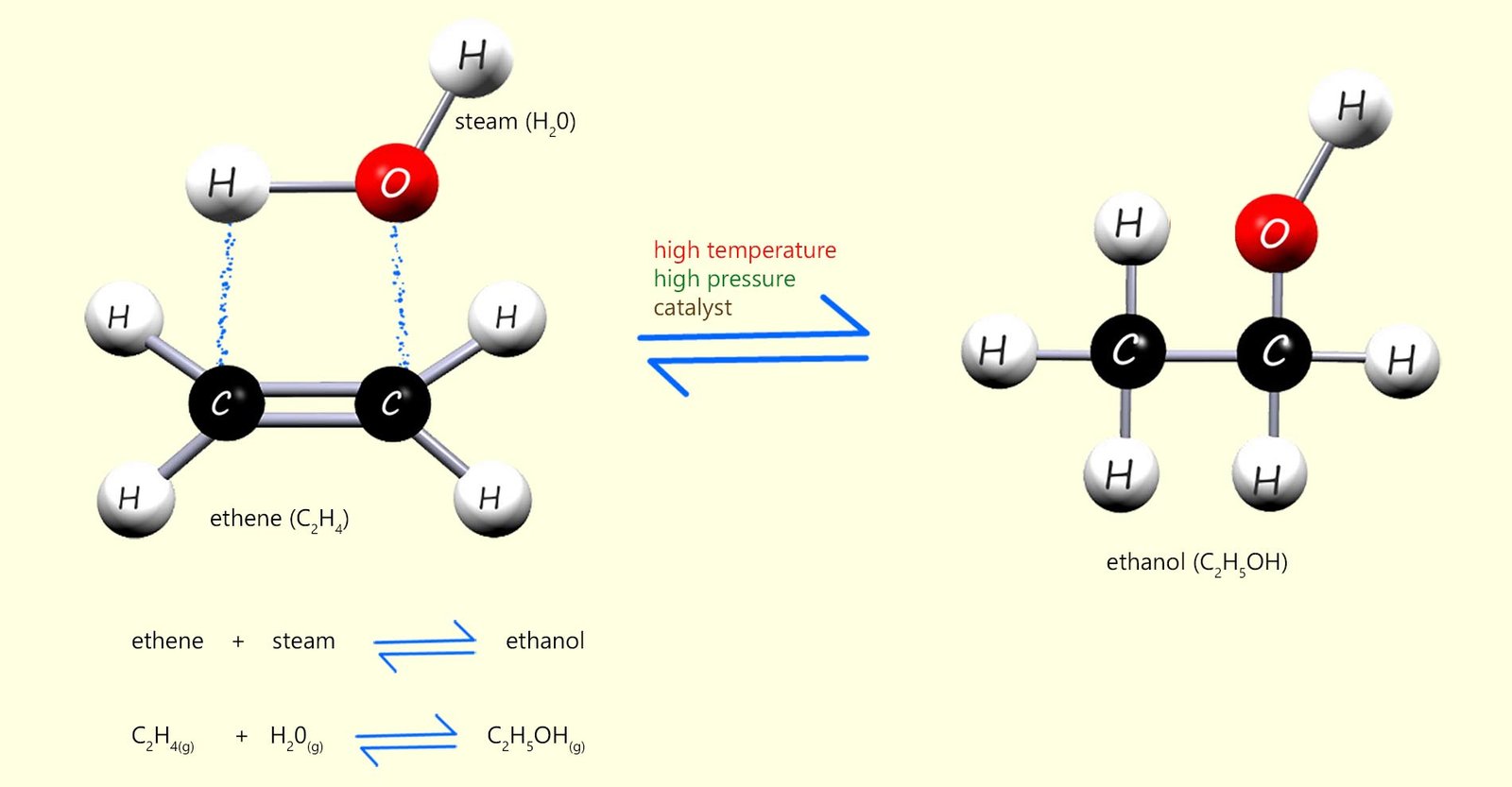
Ethene + steam (H2O)
Which product is formed?
From the equation in the image above you can see that this hydration reaction is a reversible reaction. This means that the products of the reaction will be a mixture of ethanol, steam and unreacted ethene, in fact very little of the ethene and steam react when they first enter the reaction chamber. This mixture of substances formed from the hydration reaction then leaves the reaction vessel and enters a condenser, this is outlined below:
In the condenser the steam and ethanol condense and turn back into liquids while the unreacted ethene is recycled back through the reactor to react with more steam, this way the yield of the reaction can be greatly increased. The ethanol and water formed during the reaction mix freely to form a solution which leaves the condenser. The ethanol produced by direct hydration can be separated from this solution by fractional distillation.
In fact the addition of bromine across a carbon carbon double covalent bond (C=C) is used as a test for the presence of
unsaturation in a molecule.
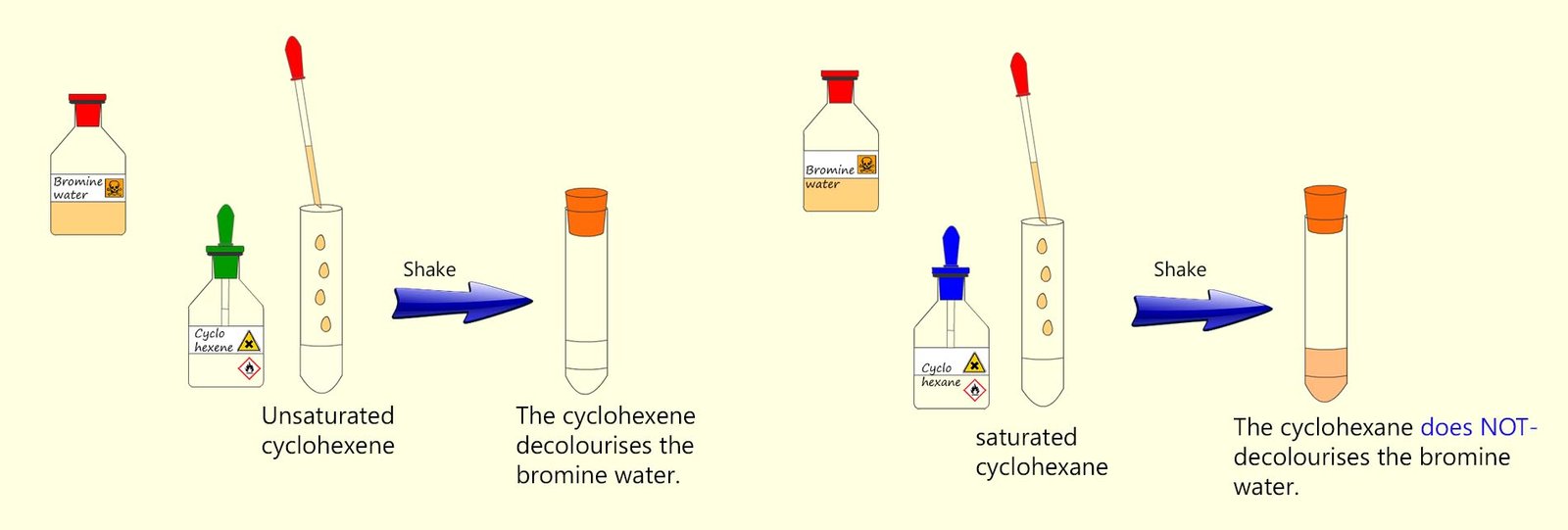
A few millilitres (ml) of liquid bromine is dissolved in water to form a red-brown solution called bromine water. When bromine water is
added to a suspected unsaturated substance
in a boiling tube and shaken then if the substance is unsaturated the bromine water will decolourise almost immediately. However if
the substance in the boiling tube is saturated and it is left in bright light then the bromine water will decolourise but very very slowly.
You may get to try out this test for unsaturation in the lab yourself. The two alkanes and alkenes that are most commonly used for this unsaturation testing are cyclohexane and cyclohexene. These are liquid alkanes and alkenes that are available in most schools. They are both clear liquids that like most organic substances do not mix with water but instead float on top of an aqueous or watery layer since they are less dense than water.
The procedure to test these two substances for the presence of a C=C double bond is as follows:
This is shown in the drawing below along with the results of the test:
In the boiling tube containing the bromine water and cyclohexane in the image above the two layers are shown coloured red orange due to the presence of dissolved bromine in each layer. However if the boiling tube is left to stand eventually any dissolved bromine in the aqueous layer will move into the organic cyclohexane layer simply because it is more soluble in the organic cyclohexane layer. This is outlined in the image below:

A very common mistake is to think that the carbon-carbon double bond stays in the product during an addition reaction. It does not.
💡 Exam trap: if the product still contains a C=C bond then it is probably wrong.
Why not us ethe summary table below to create a series of flashcards or summary notes on the reactions of alkenes?
| Reaction 🌟 | What is added 🔗 | Main product formed 🧱 | Important details to remember 🧠 |
|---|---|---|---|
| Hydrogenation | Hydrogen gas (H2) | Alkane | Turns an unsaturated alkene into a saturated alkane. Usually needs a nickel catalyst and a temperature of about 200°C. |
| Halogenation | Bromine or chlorine | Saturated halogenoalkane product | The halogen adds across the C=C bond. For example ethene + bromine forms 1,2-dibromoethane. |
| Hydration | Steam | Alcohol | Steam adds across the C=C bond. Ethene forms ethanol. Needs a phosphoric acid catalyst, about 300°C and a pressure of about 65 atmospheres. |
| Test for unsaturation | Bromine water | Colour change from orange-brown to colourless | If a molecule contains a carbon-carbon double bond, the bromine water decolourises rapidly. |
Can you tick all the statements that are correct